Den arktiske mutasjonen og Aβ-protofibriller
Professor Lars Lannfelts oppdagelse av den arktiske mutasjonen demonstrerte rollen til løselige Aβ-protofibriller i utviklingen av Alzheimers sykdom.¹
Genetisk evidens danner grunnlaget for amyloidkaskadehypotesen.
Mutasjoner i amyloidprekursorproteinet (APP) samt i presenilin 1 og 2 (PSEN1 og PSEN2), som er en del av γ-sekretasekomplekset, kan øke produksjonen eller aggregeringen av Aβ42. Disse mutasjonene forårsaker den autosomalt dominante familiære varianten av Alzheimers sykdom (ADAD), som står for mindre enn 1 % av alle Alzheimers-tilfeller.2,3 Hundrevis av slike mutasjoner er beskrevet,4 inkludert fremtredende eksempler som den svenske APP-mutasjonen (KM670/671NL) og den arktiske APP-mutasjonen (E693G).1
Den arktiske mutasjonen, identifisert i 1998 av professor Lars Lannfelt, ligger i APP-genet som koder for Aβ, og ble funnet i en svensk familie med tidlig debuterende Alzheimers. I motsetning til andre ADAD-mutasjoner øker ikke den arktiske APP-mutasjonen den totale produksjonen av Aβ. I stedet øker det tilbøyeligheten til Aβ-peptider til å aggregeres til løselige protofibriller.5–8
Til tross for denne økte aggregeringstendensen har studier vist reduserte nivåer av Aβ40 og Aβ42 i kondisjonert medium fra celler som uttrykker den arktiske APP-mutasjonen, så vel som i plasmaet til mutasjonsbærere. Denne nedgangen forklares med at mutasjonen fremmer dannelsen av protofibriller som binder Aβ til aggregerte former.9-11
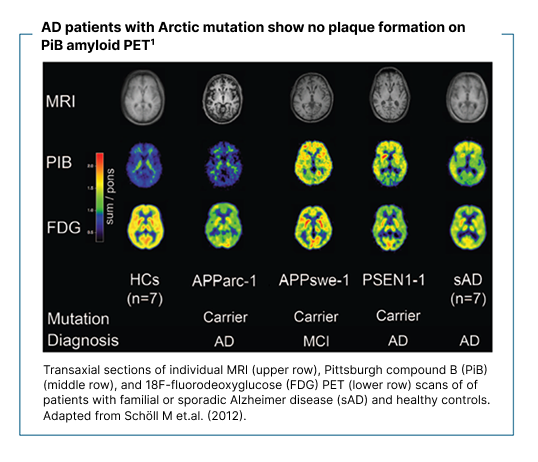
Nevropatologiske studier har vist at den arktiske APP-mutasjonen fører til utbredt Aβ-avsetning i hjernen. In vivo-avbildning med Aβ positronemisjonstomografi (PET) har imidlertid vist fravær av typisk fibrillær Aβ-plakkpatologi hos bærere og pasienter med mutasjonen, til tross for kliniske og biomarkørprofiler forenlige med Alzheimers sykdom.9–11
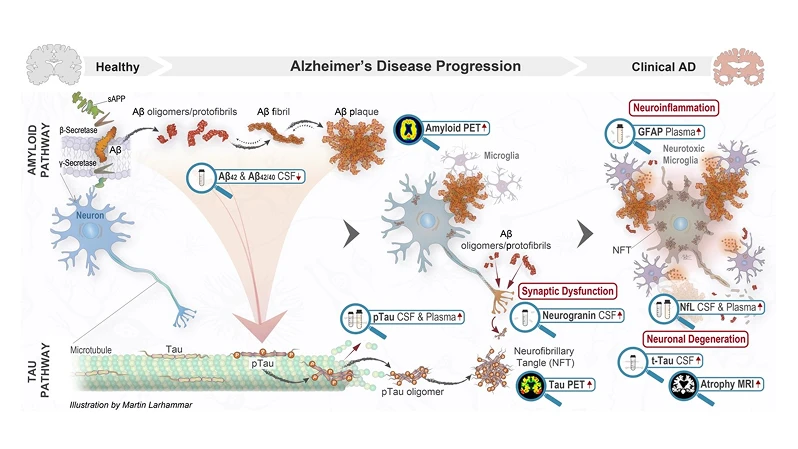
Løselige Aβ-protofibriller kan starte nedstrøms tau-patologi
Aβ-protofibriller kan initiere nedstrøms hyperfosforylering av tau-protein og dets aggregering i nevroner i form av nevrofibrillære tangler og dystrofiske nevritter.12–14 Et viktig behandlingsmål er å stoppe spredningen av tau-patologi.
Referanser
1. Nilsberth C, et al. The Arctic APP mutation (E693G) causes Alzheimer’s disease by enhanced Aβ protofibril formation. Nat Neurosci. 2001 Sep;4(9):887–93. doi:10.1038/nn0901-887
2. Goate A, et al. Segregation of a missense mutation in the amyloid precursor protein gene with familial Alzheimer’s disease. Nature. 1991 Feb 21;349(6311):704–6. doi:10.1038/349704a0
3. Sherrington R, et al. Cloning of a gene bearing missense mutations in early-onset familial Alzheimer’s disease. Nature. 1995 Jun 29;375(6534):754–60. doi:10.1038/375754a0
4. Weggen S, et al. Molecular consequences of amyloid precursor protein and presenilin mutations causing autosomal-dominant Alzheimer’s disease. Alzheimers Res Ther. 2012 Mar 28;4(2):9. doi:10.1186/alzrt107
5. Viola KL, et al. Amyloid β oligomers in Alzheimer’s disease pathogenesis, treatment, and diagnosis. Acta Neuropathol. 2015 Feb;129(2):183–206. doi:10.1007/s00401-015-1386-3
6. Johansson A, et al. Physiochemical characterization of the Alzheimer’s disease-related peptides Aβ1-42Arctic and Aβ1-42wt. FEBS J. 2006 Jun;273(12):2618–30. doi:10.1111/j.1742-4658.2006.05272.x
7. Päiviö A, et al. Unique physicochemical profile of β-amyloid peptide variant Aβ1-40E22G protofibrils: conceivable neuropathogen in Arctic mutant carriers. J Mol Biol. 2004 Jun 4;339(1):145–59. doi:10.1016/j.jmb.2004.03.004
8. Whitfield JF, et al. Alzheimer’s disease and its possible evolutionary origin: hypothesis. Cells. 2023 Jun;12(12):1618. doi:10.3390/cells12121618
9. Schöll M, et al. Low PiB PET retention in presence of pathologic CSF biomarkers in Arctic APP mutation carriers. Neurology. 2012 Jul 17;79(3):229–36. doi:10.1212/WNL.0b013e31825fdf18
10. Kalimo H, et al. The Arctic AβPP mutation leads to Alzheimer’s disease pathology with highly variable topographic deposition of differentially truncated Aβ. Acta Neuropathol Commun. 2013 May 10;1:60. doi:10.1186/2051-5960-1-60
11. Basun H, et al. Clinical and neuropathological features of the Arctic APP gene mutation causing early-onset Alzheimer disease. Arch Neurol. 2008 Jan;65(4):499. doi:10.1001/archneur.65.4.499
12. Hampel H, et al. The amyloid-β pathway in Alzheimer’s disease. Mol Psychiatry. 2021 Oct;26(10):5481–5503. doi:10.1038/s41380-021-01249-0
13. He Z, et al. Amylod-β plaques enhance Alzheimer’s brain tau-seeded pathologies by facilitating neuritic plaque tau aggregation. Nat Med. 2018 Jan;24(1):29–38. doi:10.1038/nm.4440
14. Lewis J, et al. Enhanced neurofibrillary degeneration in transgenic mice expressing mutant tau and APP. Science. 2001 Aug 24;293(5534):1487–91. doi:10.1126/science.1058189